- Research Overview
- Current Research
-
Previous Work
- Polyphenism Evolution: Phenotypic Plasticity and Locomotor Performance in Spadefoot Toads
- Life History Evolution: Costs of Reproduction, Offspring Size/Number Trade-Offs, and Locomotor Performance
- Morphological Scaling Relationship Evolution: Butterflies
- Ecological & Functional Morphology
- Development & Trait (Micro) Evolution
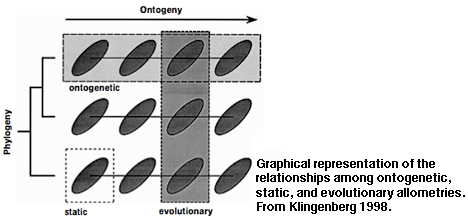
A Primer on a Multidisciplinary Approach to the Study of Animal Form
“The most pressing need is to develop methods of distinguishing the relative roles the ‘external’ (e.g., ecological sources of selection) and ‘internal’ factors (e.g., genetic correlations, developmental constraints) play in determining evolutionary rates.”. — From the 2001 joint report defining the challenges facing the science of evolution published by the American Society of Naturalists, Society for the Study of Evolution, Society for Molecular Biology and Evolution, Ecological Society of America, Society of Systematic Biologists, Genetics Society of America, Animal Behavior Society, and Paleontological Society. (Meagher TR, Futuyma DJ. Executive Document: Evolution, Science, and Society. The American Naturalist 2001;158(S1):1-45)
Diversity in animal morphology has evolved largely through changes in the relative size of morphological traits. For example, kangaroos possess forelimbs several times smaller than their hindlimbs whereas orangutans have greatly elongated arms relative to their legs. Such changes in the relative size of morphological traits produce much of the diversity of animal form present among orders, families, genera, species, populations, alternative morphs, and even between the sexes. Yet within such animal groups, individuals usually exhibit similarly proportioned parts, such that the size of limbs, wings, teeth, or other body parts are properly sized relative to each other and to the rest of the body.
Over 75 years ago, Julian Huxley formalized the study of scaling relationships between traits, which he called ‘allometries’, with a general power equation describing the relative growth of two organs through time or the relative size of two traits across biological groups such as species. Armed with this equation, biologists described the scaling relationships for numerous traits among many animal groups in an effort to understand how changes in allometries relate to diversity in animal form; this diversity is captured by variation among groups in the slopes and intercepts of the allometric equation.
Appreciating the importance of size and discovering regularities among scaling relationship, however, does not explain how scaling relationship parameters develop or evolve. Such explanations have been offered by quantitative genetic theory, and are also a product of emerging developmental and physiological-based models of trait evolution that focus on the mechanisms regulating and coordinating trait growth and size covariation. Recent advances in developmental biology, morphometrics, and statistical treatments of functions (function-value traits) provide new opportunities to evaluate the predictive power of these models, and to understand how the proximate mechanisms that produce scaling relationships influence allometry evolution. At the other end of the spectrum, a variety of phenotypic engineering or other manipulative approaches can be used now to estimate the form and strength of selection acting on these multivariate traits. An integrative research program that blends methodology from comparative biology, quantitative genetics, developmental genetics, functional morphology, and evolutionary ecology promises for the first time to elucidate how internal developmental processes and external natural selection shape the evolution of scaling relationships – in other words, how these forces shape the evolution of morphology itself.
Despite the fact that scaling relationships are central to the evolution of animal form, physiology, life history, etc., the study of these relationships can be quite mathy, dry, and sometimes seem uninteresting on the surface. One way to expore the rich topic of scaling in a fun way is to think about scaling in B-monster movies from the 1950s. Michael C. LaBarbera at the University of Chicago has a great page focusing on on just this topic; from his page, “Size has been one of the most popular themes in monster movies … The premise is invariably to take something out of its usual context—make people small or something else (gorillas, grasshoppers, amoebae, etc.) large—and then play with the consequences. However, Hollywood’s approach to the concept has been, from a biologist’s perspective, hopelessly naïve. Absolute size cannot be treated in isolation; size per se affects almost every aspect of an organism’s biology.”
In my current work, I take an integrative approach to study morphological scaling relationship evolution using a combination of artificial selection and fitness assays as described here, using the fruit fly Drosophila melanogaster as an empirical model. D. melanogaster is the flagship animal model of population genetics, development and genomics. Moreover, one of the most thoroughly studied of all morphological traits is the Drosophila wing, which is a model for the study of growth regulation. Moreover, fly wings exhibits interesting, unexplained patterns of variation in shape and relative size, and are ecologically important – being used for both flight and courtship song production. I use wing shape and relative wing size as empirical models of complex trait suite expression and evolution in several complementary studies, described briefly below. As a postdoctoral researcher, I studied the evolution of morphological scaling relationships as described in detail here.
Relevant publications
Dreyer, A.P., O. Saleh Ziabari, E.M. Swanson, A. Chawla, W.A. Frankino, A.W. Shingleton. 2016 Cryptic individual scaling relationships and the evolution of morphological scaling. Evolution 70:1703-1716.
Mirth, C.K., W. A. Frankino and A.W. Shingleton. 2016. Allometry and Size Control. invited submission. Current Opinion in Insect Science 13:93-98.
Stillwell, R.C., A.W. Shingleton, I. Dworkin and W.A. 2016. Frankino. Tipping the scales: Evolution of the allometric slope independent of average trait size. Evolution 70:433-444.
Shingleton, A.W. and W. A. Frankino. 2013. New perspectives on the evolution of exaggerated traits. BioEssays 35:100-107.
Stillwell, R. C., I. M. Dworkin, A. W. Shingleton, W. Anthony Frankino. 2011. Experimental manipulation of body size to estimate morphological scaling relationships in Drosophila. Journal of Visualized Experiments (56) e3162.
Frankino, W. A., A. Shingleton, D. Emlen. 2009. Experimental approaches to studying the evolution of morphological allometries: The shape of things to come. in: Experimental Evolution: Concepts, Methods, and Applications, T. Garland and M. Rose, eds. University of California Press.
Frankino, W. A., B. J. Zwaan, D. L. Stern, and P. M. Brakefield. 2007. Internal and external constraints in the evolution of a forewing-hindwing allometry. Evolution 61:2958-2970.
Shingleton, A., W. A. Frankino, T. Flatt, F. Nijhout, and D. Emlen. 2007. Developmental mechanisms and the evolution of allometries. BioEssays 29:536-548.
Frankino, W. A., D.S. Stern, and P.M. Brakefield. 2005. Developmental constraints and natural selection in the evolution of allometries. Science 307:718-720.